Why this topic matters right now
The conversation around insulin is louder than ever before, not only in the United States but also across Canada and other parts of the world. People with diabetes depend on insulin every day to survive, and yet many face a constant financial burden. In the U.S., where the average cost of a vial of insulin can be five to ten times higher than in peer countries, patients often ration their doses, switch to cheaper but less suitable alternatives, or risk dangerous gaps in their treatment. This reality has forced many families to ask: Why is insulin so much cheaper in Canada, and what do I need to know before considering buying it there?

Beyond the cost issue, Canada itself is undergoing significant changes in how insulin and other prescription drugs are regulated and covered. With discussions around a national pharmacare program and provinces shifting toward biosimilars, patients—even within Canada—are experiencing new challenges and opportunities when it comes to accessing their life-saving medication. For Americans, this raises more questions: if I cross the border or order online, am I safe? Is it legal? Will I really get the same product my doctor prescribed?
This article aims to cut through the confusion. Instead of vague “insulin is cheaper in Canada” headlines, we’ll dig into the details: how insulin is approved, why brand names differ, what legal loopholes exist for importing, how storage and labeling rules change, and the myths you’ve probably heard. By the end, you’ll understand not only the truth about Canadian insulin but also how to make safer, more informed decisions—whether you’re a Canadian navigating policy shifts or an American considering a cross-border purchase.
What “Canadian insulin” actually means
When people say “Canadian insulin,” they often imagine some completely different drug—like it’s a special brand made only in Canada. In reality, insulin in Canada goes through a rigorous process of review by Health Canada, the federal regulator responsible for ensuring safety, quality, and effectiveness. Much like the FDA in the U.S., Health Canada won’t authorize an insulin product for sale unless the clinical evidence shows it works as expected, the manufacturing meets strict quality standards, and the labeling clearly communicates risks and instructions.
A unique feature of Canadian drug regulation is the DIN (Drug Identification Number). Every insulin product sold in Canada—whether a vial, cartridge, or pen—carries an eight-digit DIN. This number isn’t just a bureaucratic stamp. It’s a way to confirm that the product is legitimate and approved. For example, if you buy insulin online or at a Canadian pharmacy, you can look up the DIN in Health Canada’s Drug Product Database and verify that it matches the authorized product. This safeguard helps patients avoid counterfeits, which are unfortunately a rising risk in the age of online shopping.
Another important distinction is the way Canada handles biologics and biosimilars. Insulin is classified as a biologic—meaning it’s made using living cells, which makes it more complex than traditional chemical drugs. Canada has approved biosimilar insulins (essentially “generic-like” versions of originator insulins), but unlike in the U.S., Health Canada doesn’t decide on interchangeability. That power lies with provincial drug plans. So, while a biosimilar might be approved nationally, whether you can be automatically switched at the pharmacy counter depends on where you live. This is one reason why you might hear different stories from Canadians in different provinces.
Is Canadian insulin the same as U.S. insulin?
This is probably the single most common question Americans ask. The short answer: yes, the molecule is usually the same, but the brand name, packaging, or device may be different. Take insulin aspart, for example. In the U.S., most patients know it as NovoLog, while in Canada it’s marketed as NovoRapid. Both contain the same active ingredient, both lower blood sugar in the same way, and both are manufactured under high-quality standards. But the box, instructions, and sometimes even the inactive ingredients may differ slightly.
That difference in branding can create confusion. Imagine a traveler with diabetes being prescribed NovoLog in the U.S. but suddenly handed a box of NovoRapid in Canada. Without prior knowledge, they might think it’s a completely different drug. This is why it’s so important to double-check labels, DIN numbers, and your doctor’s instructions. Other examples include Lantus (insulin glargine U-100), which exists under the same name in both countries, versus Fiasp (faster-acting insulin aspart), which may be marketed slightly differently.
It’s also worth noting that insulin strengths vary. While U-100 remains the most common concentration in Canada, you’ll also find U-200 and U-300 pens (such as Tresiba and Toujeo). If you’re used to one strength in the U.S. but grab a different one in Canada, it’s easy to make dosing errors. Pens, cartridges, and syringes are designed for specific strengths, and mixing them up can lead to under- or overdosing. So while the molecule may be the same, the real-life usage might not be if you’re not careful.
Prices and why they’re different
The elephant in the room is price. Why can a vial of insulin in the U.S. cost $200–$300, while in Canada the same product might cost under $50? The answer lies in how each country regulates drug prices. Canada has a federal body called the Patented Medicine Prices Review Board (PMPRB), which evaluates whether a drug’s price is “excessive” compared to peer countries. While manufacturers can propose a price, they can’t set it sky-high without triggering review or rejection. Provinces also negotiate bulk prices with manufacturers, which drives costs down even further.
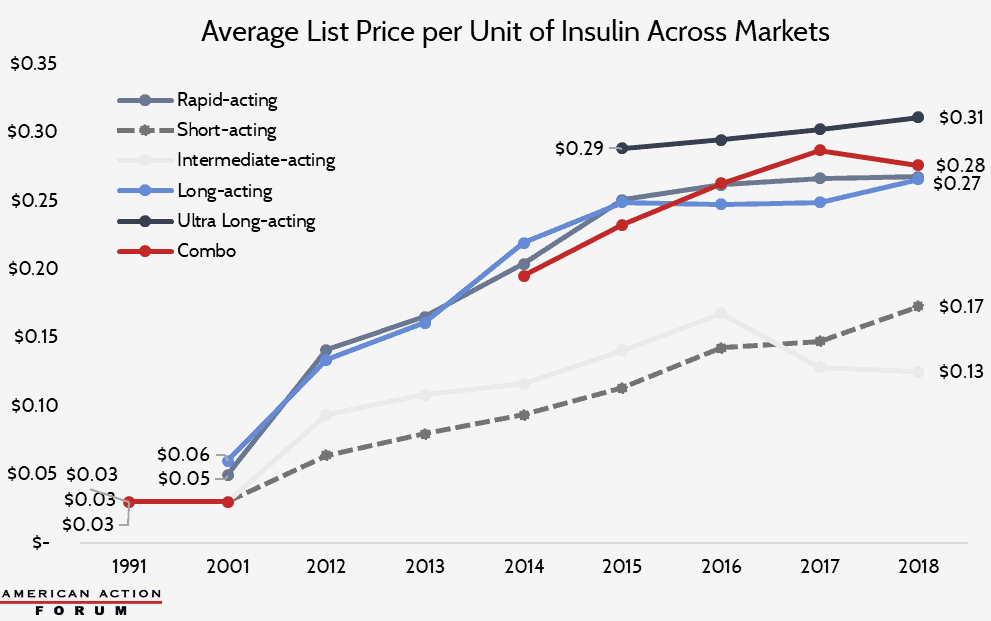
By contrast, the U.S. system relies heavily on negotiations between drug manufacturers, insurers, and pharmacy benefit managers (PBMs). These negotiations often involve complex rebates and discounts, but the “list price” remains high. Patients without good insurance—or those stuck in high-deductible plans—end up paying the most. That’s why even though insulin manufacturers announced price caps in recent years, the actual out-of-pocket cost in the U.S. can still vary wildly depending on coverage.
Looking ahead, Canada is introducing pharmacare reforms that could shift the insulin landscape even more. In early 2024, the federal government announced plans for a national pharmacare program with insulin among the first drugs to be covered. If provinces sign on, this could mean even lower costs and broader access for Canadians, potentially widening the affordability gap between Canada and the U.S. That makes it even more likely that Americans will keep looking north for relief.
Can Americans buy insulin from Canada? The legal fine print
This is where things get tricky. Under U.S. law, importing prescription drugs from abroad is generally illegal. The FDA does have a Personal Importation Policy (PIP), which gives them discretion not to enforce the rule in some cases—usually if the patient brings in less than a 90-day supply, the drug isn’t considered a significant health risk, and it’s clearly for personal use. But the keyword here is discretion. There’s no guarantee that your insulin won’t be stopped at the border.
If you’re traveling to Canada, you might assume you can simply walk into a pharmacy and stock up. In reality, you’ll need documentation. Many Canadian pharmacies will ask for a prescription, especially if you’re not a Canadian resident. Even if insulin is technically “behind-the-counter” in some provinces, pharmacists are required to keep records and ensure safe dispensing. And when you return to the U.S., Customs and Border Protection (CBP) officers can still question or confiscate your supply. That risk is important to understand before making any decisions.
Another concern is legitimacy. Not every website that claims to be a Canadian pharmacy is actually based in Canada. Some ship drugs from India, Turkey, or other countries under the guise of “Canadian pricing.” To stay safe, always verify that the pharmacy is licensed by a Canadian provincial regulatory body and that the product has a valid DIN number. Reputable pharmacies will also provide pharmacist counseling, not just ship drugs blindly. Skipping these checks could mean ending up with unsafe or counterfeit insulin, which is the last thing any diabetic needs.
Biosimilar switching policies by province
One of the biggest differences between Canada and the U.S. when it comes to insulin is how biosimilars are introduced. In Canada, each province and territory controls its own public drug plan, which means that policies around biosimilar insulin differ depending on where you live. Provinces like British Columbia, Alberta, and Ontario have already launched Biosimilars Initiatives that require patients to switch from originator biologics (such as Humalog or Lantus) to their biosimilar equivalents. This is usually done in phases, giving patients and providers time to adjust.
For patients, the idea of switching can feel intimidating. After all, insulin is not like a simple tablet—you’re injecting a biologic drug that keeps you alive. But Health Canada assures patients that biosimilars are just as safe and effective as originators, having passed the same rigorous approval process. The main difference is cost: biosimilars are significantly cheaper, which is why provinces are encouraging or even mandating the switch to keep public healthcare sustainable.
Still, switching isn’t as simple as swapping a pill bottle. Devices, pen compatibility, and insurance coverage all play a role. For example, if you’re using a pump or have a specific pen system, moving to a biosimilar might mean retraining on a new device. Some patients have reported feeling anxious about the transition, but provincial health ministries have created support programs to help with counseling, education, and follow-up. Knowing the local policy is key—because what applies in British Columbia may not be the same in Ontario.
Traveling with insulin in/through Canada
Travel can be stressful enough without worrying about how to transport insulin safely. If you’re flying within or through Canada, the Canadian Air Transport Security Authority (CATSA) has clear guidelines: insulin, needles, syringes, pumps, and continuous glucose monitors (CGMs) are allowed in your carry-on bag. The golden rule is simple—always carry insulin in your hand luggage, never in checked baggage. Cargo holds can get too hot or too cold, which can ruin insulin.
Another tip is to keep everything clearly labeled. Having your insulin in its original packaging with a prescription label makes things easier at airport security checkpoints. While a doctor’s note isn’t always required, it can save you time if agents have questions. And if you use an insulin pump or CGM, be prepared to explain how it works—security officers are trained to recognize these devices, but extra clarity can speed up the process.
Temperature is another major factor. Most insulin vials and pens need to be stored in the fridge until first use, but can remain at room temperature for a set period afterward. For example, some insulins last up to 28 days, while others, like Toujeo, can last up to 8 weeks once opened. Portable cooling cases or gel packs can be lifesavers on long trips. If you’re crossing multiple time zones, also consider how to adjust your injection schedule—you may need to consult your doctor beforehand to avoid dosing mistakes while traveling.
Units, meters, and labels: mmol/L vs. mg/dL
One often-overlooked difference between Canada and the U.S. is blood sugar units. In the U.S., most patients are familiar with mg/dL (milligrams per deciliter). In Canada, however, blood sugar is measured in mmol/L (millimoles per liter). At first glance, this might seem like a minor detail, but it can lead to dangerous misunderstandings if you’re not careful. Imagine a U.S. patient with a target blood sugar of 100 mg/dL. If they confuse units and think “100 mmol/L” is normal, they’d actually be looking at an off-the-charts reading (equivalent to 1800 mg/dL!).
The good news is that the math is straightforward: mg/dL ÷ 18 ≈ mmol/L. For example, 180 mg/dL equals about 10 mmol/L. Conversely, mmol/L × 18 ≈ mg/dL. Most modern glucose meters can switch between units in their settings, but you need to double-check before using one in a new country. If you bring your own meter to Canada, keep it in your familiar units to avoid confusion. If you buy supplies there, confirm with your healthcare provider that the numbers align with your treatment plan.
Labels can also vary. Canadian insulin packaging will display DIN numbers and instructions in both English and French, while U.S. packaging typically uses English only. Patient information leaflets may be worded differently, which is why it’s smart to read the Canadian leaflet carefully—even if you think you already know the product. A small difference in instructions, timing, or storage advice could matter more than you realize.
Safety checklist before using insulin sourced in Canada
If you’re considering using Canadian insulin, safety has to come first. Step one is making sure you’re actually using the right type of insulin. Is it rapid-acting, short-acting, intermediate, or long-acting? Switching from Humalog (lispro) to NovoRapid (aspart) without guidance could throw off your meal planning, timing, or pump settings. Even if the insulin molecule is the same, pen devices may differ in dose increments or delivery methods.
Step two is verifying the strength. U-100 is standard, but if you accidentally pick up a U-200 or U-300 insulin pen, your doses could be double or triple what you intended. Never assume pens are interchangeable. Double-check the label, the concentration, and the instructions. If you’re unsure, call the pharmacy before using it.
Finally, look at the DIN, expiry date, and packaging. If anything seems off—like a smudged label, missing DIN, or unfamiliar-looking packaging—don’t use it until you verify with Health Canada’s database or a licensed pharmacist. Counterfeit drugs do exist, and insulin isn’t something you can take chances with. And of course, involve your healthcare team before making any switch. Even small differences can have a big impact on your blood sugar control, so keeping your doctor in the loop is crucial.
Common myths—busted
“Canadian insulin is lower quality.” This is one of the most persistent myths, and it couldn’t be further from the truth. Insulin sold in Canada has to pass the same rigorous standards of manufacturing and quality control as any other biologic drug. Health Canada requires detailed clinical evidence, quality inspections, and post-market surveillance to ensure safety. In fact, some of the same factories that supply Canadian insulin also supply the U.S. market.
“It’s always the same pen as in the U.S.” Not necessarily. While the insulin molecule may be identical, the devices and instructions may differ. For example, a pen sold in the U.S. may be branded differently or have slightly different instructions than the Canadian version. Patients should never assume 1:1 interchangeability. Always confirm with your doctor or pharmacist how to use the new device correctly.
“You don’t need a prescription for insulin in Canada.” This one is a half-truth. In some provinces, insulin is classified as Schedule II, which means it can be dispensed by a pharmacist without a doctor’s prescription—but it’s still kept behind the counter. The pharmacist has to record the transaction, provide counseling, and ensure safe use. That’s very different from grabbing insulin off a grocery store shelf. And for non-residents, most pharmacies will still ask for a prescription, especially for safety and liability reasons.
Who benefits most—and who should be extra careful
For Canadians, the biggest benefit of these policies is financial. With public drug plans covering more biosimilars and pharmacare proposals on the horizon, many patients will see their costs go down. For Americans, the primary benefit is affordability—if they can access Canadian insulin legally and safely. U.S. patients with no insurance or those stuck paying high out-of-pocket costs are the ones most motivated to look north.
That said, not everyone is an ideal candidate for switching. People using insulin pumps or advanced hybrid closed-loop systems need to be especially careful. These devices are calibrated for specific insulin types and strengths, so even a small change in formulation can throw off the system. Pregnant women, children, and patients with complicated cases of Type 1 diabetes also need close supervision before changing brands or sources of insulin.
The takeaway is that Canadian insulin can be a lifeline for many—but it’s not a one-size-fits-all solution. If you’re considering it, weigh the benefits against the risks, and involve your healthcare team in every step.
How to talk to your healthcare team
One of the best steps you can take is to have an open conversation with your doctor or diabetes educator. Many patients feel nervous admitting that they’re considering buying insulin from Canada, but hiding it only increases the risk. Your care team can help you figure out whether the Canadian version of your insulin is equivalent, how to adjust doses if needed, and what to watch for during the transition.
Come prepared with questions. For example: “If I have to switch from NovoLog to NovoRapid, do I change my timing?” or “How do I make sure a U-200 pen is safe if I’ve only used U-100 before?” Bring your current devices and prescriptions with you so your doctor has all the context. And if you’re traveling, ask about a written care plan in case of emergencies.
Documentation is your friend. Having printed prescriptions, device manuals, and a summary of your dosing schedule can save you a lot of trouble if you run into issues abroad. It also helps your doctor give more specific advice, instead of guessing based on memory. The more transparent you are, the safer your switch or travel will be.
Key takeaways
Canadian insulin isn’t a mysterious or inferior version of the drug—it’s regulated, safe, and often the very same molecule sold in the U.S., just under a different name. The biggest differences are in price, branding, devices, and regulations. Canadians benefit from stricter price controls, while Americans face legal and logistical hurdles when trying to access cheaper options across the border.
The key points to remember: always check the DIN, verify the insulin type and strength, and involve your healthcare provider before switching. Be aware of biosimilar substitution policies if you live in or travel through different provinces, and don’t underestimate the importance of units (mmol/L vs. mg/dL). And most importantly, never make assumptions. What looks like the “same” pen may not be used the same way.

Whether you’re a Canadian navigating provincial biosimilar policies or an American weighing the risks of cross-border insulin, knowledge is your most powerful tool. With the right preparation, you can make safer, smarter, and more affordable choices about your diabetes care.
Conclusion
“Canadian insulin” isn’t a shortcut so much as a different system—with its own rules, labels, prices, and policies. If you understand how Health Canada authorizes biologics, how brand names and strengths line up, how biosimilar switching works provincially, and what cross-border rules actually allow, you can make safer, smarter decisions. Whether you’re a Canadian navigating plan changes or a U.S. patient comparing options, the winning strategy is the same: verify the product (DIN), match the strength/device, and loop in your clinician—especially when switching brands or traveling.
FAQs
1) Is there a once-weekly insulin in Canada?
Yes. Insulin icodec (Awiqli) received Health Canada approval in 2024 for adults with diabetes. Provincial formulary listings and coverage vary
2) If I buy insulin in Canada, will TSA or CATSA take it away?
Diabetes supplies (insulin, needles, pumps, CGMs) are permitted in carry-ons. Keep items labeled, bring a prescription/letter, and allow extra time at security.
3) Can I switch from Lantus to a biosimilar on my own?
Don’t switch solo. Provinces may mandate biosimilar transitions, but you should coordinate with your prescriber to confirm dose, device, and timing.
4) Why does my Canadian meter show mmol/L instead of mg/dL?
Canada uses mmol/L. Convert by dividing mg/dL by 18 (or multiplying mmol/L by 18). Ensure your meter is set to the units your team uses.
5) Do I need a prescription to buy insulin in Canada?
In several provinces, insulin is classified as Schedule II—dispensed by a pharmacist without a physician’s prescription but kept behind the counter with documentation and counseling. Policies are provincial; carry your prescription when traveling.